When Negative Cultures Lie: The Hidden Cost of Diagnostic Blind Spots
When a culture comes back negative, most clinicians take it at face value. But a negative result is only as reliable as the method used to obtain it.

When a culture comes back negative, most clinicians take it at face value: no pathogen detected, no infection present. It's a reasonable assumption, until it isn't.
On the latest episode of Prescribe Responsibly, I spoke with Tami Burdick, a patient advocate who spent seven months navigating a rare breast infection caused by Corynebacterium kroppenstedtii. The organism was there the entire time. The lab just couldn't see it.
The Organism That Hides in Plain Sight
Corynebacterium kroppenstedtii is not a household name, even among clinicians. But it is increasingly recognized as a causative agent in granulomatous mastitis, a chronic inflammatory breast condition that mimics cancer on imaging and confounds traditional diagnostic methods. The challenge is that this organism does not reliably grow in standard cultures. It is not always visible on a Gram stain.
"These Gram stains and cultures often fail to detect this opportunistic microorganism," Burdick explains during our conversation on Prescribe Responsibly. "It doesn't always grow in a culture. It's not always seen on a Gram stain."
In Tami's case, an early culture grew diptheroids, a finding her infectious disease specialist dismissed as contamination or normal skin flora. But diptheroids have a strong correlation with Corynebacterium, a genus with over 80 strains, many of which are pathogenic. That dismissed finding was a direct clue to the causative organism.
The False Negative Trap
This case illustrates a broader diagnostic truth that extends well beyond rare breast infections. In infectious disease, a negative result is only as reliable as the method used to obtain it. When clinicians treat a negative Gram stain or culture as definitive evidence that no pathogen is present, they fall into what I call the false negative trap.
The consequences are real. In Tami's case, the missed organism led to months of delayed treatment, disease progression, two breast abscesses connected by sinus tracts, and ultimately surgery that left permanent scarring. Had the initial diptheroids finding been pursued with molecular methods — PCR, or next-generation sequencing, the causative organism might have been identified in weeks, not months.
"It's not really the patient's responsibility to hunt down labs and to see what labs are available," Burdick says. But in the absence of clinician awareness, that's exactly what she was forced to do. The diagnostic test that finally identified her organism was one she found through her own research — and the post-surgery confirmation had to be sent to the Mayo Clinic.
Why Clinicians Don't Order What They Don't Know Exists
The diagnostic gap is not a failure of intelligence. It's a failure of infrastructure. Tami identifies several contributing factors from her journey through five physicians:
Unfamiliarity with advanced testing. A surgical breast oncologist cannot be expected to know every molecular infectious disease test available for rare organisms. That knowledge often sits with infectious disease specialists — yet many patients with granulomatous mastitis never see one.
Outdated training. Clinicians may be relying on what they learned in medical school decades ago. The science around Corynebacterium and its pathogenic potential has evolved significantly, but continuing education has not always kept pace.
Expense concerns. Non-traditional microbiology tests can be costly, and insurance coverage varies. Clinicians may hesitate to order tests they're unsure will be covered or clinically justified.
The volume problem. Burdick's surgeon contributed a candid admission to her book: she simply did not have the time — due to her high clinical load — to do the research that Tami did. When physicians manage large patient volumes with minutes per case, rare diagnoses fall through the cracks.
Precision as Infrastructure
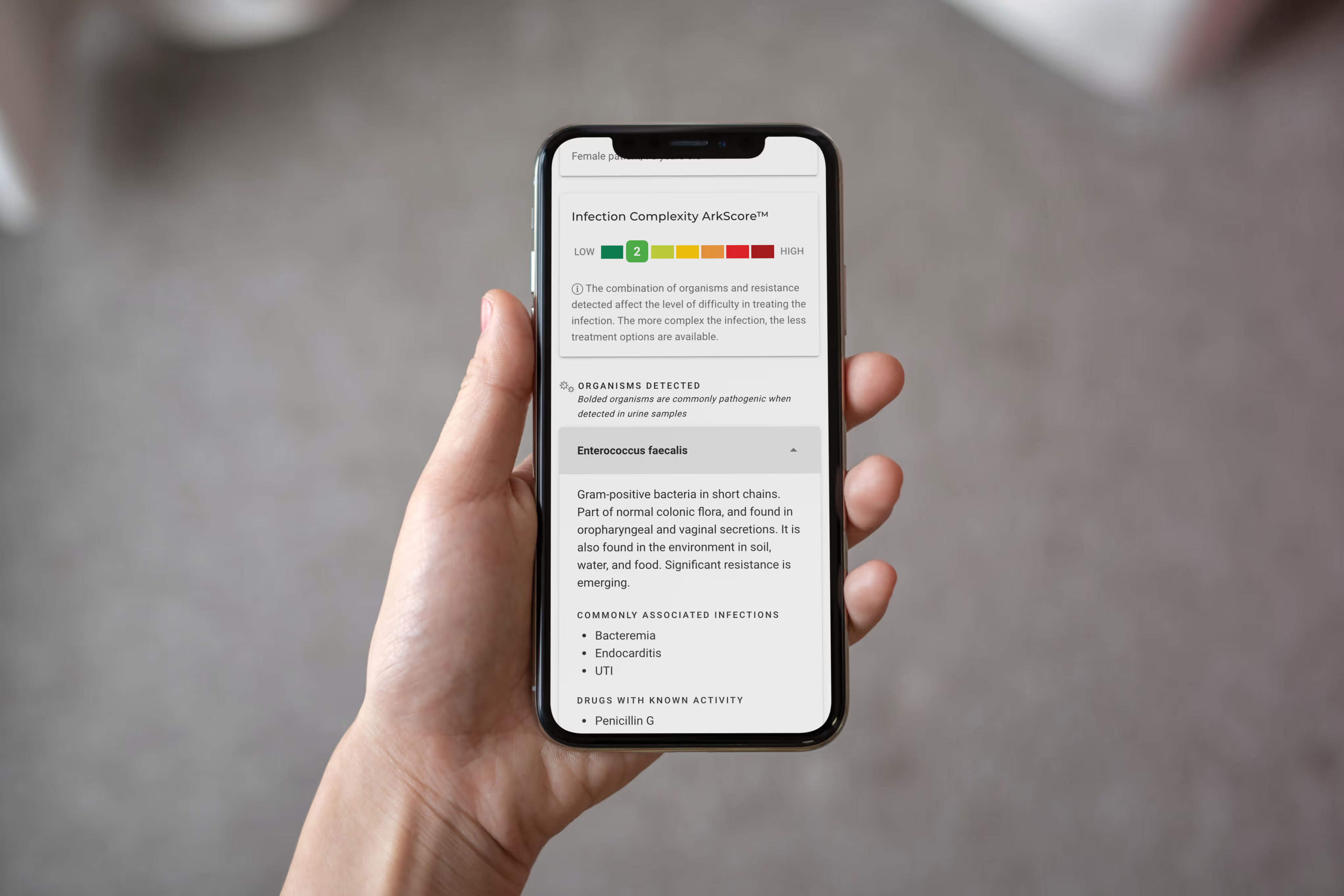
This is where clinical decision support becomes not a luxury, but a necessity. When a clinician receives a lab report that says "no growth," that should not be the end of the diagnostic story — especially when clinical presentation suggests otherwise. Tools like Arkstone's OneChoice can interpret molecular data, flag organisms that conventional cultures miss, and guide clinicians toward the right next step.
The goal is not to replace the clinician's judgment. It's to ensure that a negative culture prompts the right question: "What might we be missing?"
For the laboratory, the implication is equally significant. In a market where every lab offers the same types of tests, the lab that provides intelligent interpretation becomes a partner in patient care, not just a vendor of results.
The Patient Shouldn't Have to Be the Expert
Tami Burdick did extraordinary work to diagnose her own condition. She researched the tests, advocated for them with her surgeon, and traced the environmental source of her infection to contaminated well water in Connecticut. That kind of initiative saved her life. But it shouldn't have been necessary.
The future of infectious disease diagnostics is one where no patient has to become a detective to receive a correct diagnosis. That future requires labs that go beyond culture, clinicians who have access to decision support, and a system that treats "no growth" as a data point — not a final answer.
🎧 Listen to the full conversation with Tami Burdick on Prescribe Responsibly, Episode 2 — available on Spotify, Apple Podcasts, and YouTube.
This content is for educational purposes only and does not constitute medical advice.
Share this article
Blogs
Latest Blogs
Stay informed with our featured blogs.

When Negative Cultures Lie: The Hidden Cost of Diagnostic Blind Spots
When a culture comes back negative, most clinicians take it at face value. But a negative result is only as reliable as the method used to obtain it.
When a culture comes back negative, most clinicians take it at face value. But a negative result is only as reliable as the method used to obtain it.

When Antibiotics Miss the Mark Prescribe Responsibly · Episode 2
A conversation with Tami Burdick, Patient Advocate and Author of Diagnosis Detective: Curing Granulomatous Mastitis.
A conversation with Tami Burdick, Patient Advocate and Author of Diagnosis Detective: Curing Granulomatous Mastitis.
How Arkstone saves healthcare payers billions by eliminating unnecessary prescriptions
Most patients never know how often the right antibiotic is a guess. A faster, more confident answer changes everything for the patient, the clinician, and the hospital.
Most patients never know how often the right antibiotic is a guess. A faster, more confident answer changes everything for the patient, the clinician, and the hospital.
Customer Testimonials
Arkstone's solutions have transformed our patient care.
Take the Next Step, Try OneChoice®
No more guesswork. No more lookup tools. No more inaccuracies and inconsistencies. No more stopping to ask for directions.