When Antibiotics Miss the Mark Prescribe Responsibly · Episode 2
A conversation with Tami Burdick, Patient Advocate and Author of Diagnosis Detective: Curing Granulomatous Mastitis.

The Patient Who Solved Her Own Case
Tami Burdick woke up one morning with pain in her breast. What followed was not a swift diagnosis and targeted treatment. It was a seven-month odyssey through five physicians, a misinterpreted culture, two abscesses, sinus tracts tunneling through tissue, and a wound vac she wore for six weeks. Along the way, her presentation had the appearance of what could be cancer, and culture results were almost dismissed entirely, all because the organism behind her infection was one that conventional lab methods routinely miss.
Her diagnosis: granulomatous mastitis, a rare, chronic inflammatory breast condition. Her underlying cause: a bacterial infection with Corynebacterium kroppenstedtii, contracted from contaminated water during a business trip to Connecticut. But it took Tami, along with her doctors, to piece that together. A collective effort.
On this episode of Prescribe Responsibly, Dr. Ari Frenkel sits down with Tami to explore what happens when infections are poorly understood, organisms go undetected, and antibiotics become a trial-and-error process. The conversation is a window into the human cost of diagnostic failure, and a case study in why precision matters.
Five Doctors and a Mammogram Rated for Cancer
Tami's journey began with a self-breast exam that revealed a lump and hardened area. Her primary care physician ordered a mammogram that came back BI-RADS 5 — highly suspicious for malignancy. Before the biopsy was even performed, she was referred to a surgical breast oncologist. Tami was preparing for a cancer diagnosis.
"I just flat out said to the radiologist: I'm expecting and prepared for a breast cancer diagnosis. He said, 'While this very well could be cancer, I'm not thinking that's what this is. But whatever this is, it's chronically inflamed, and that's what you need to figure out. Why?'"
That conversation became Tami's mission. Over the following months, she would see five doctors: her PCP, a radiologist, a surgical breast oncologist, an infectious disease specialist, and a functional medicine practitioner.
The Clue That Was Dismissed
The first critical misstep came early. Tami's initial culture grew diphtheroids, a finding her infectious disease specialist dismissed as contamination or normal skin flora. The specialist told her she could do antibiotics if she wanted, but didn't have to.
"Why would I do antibiotics if I don't have to?" Tami recalls thinking, aware of the risks of unnecessary antibiotic exposure. But what she didn't know at the time was that diphtheroids have a strong correlation to Corynebacterium — and there are over 80 strains, many of which are pathogenic.
That dismissed finding was a direct clue to the organism that was causing her disease. Had it been investigated further, Tami's seven-month journey might have been weeks.
The disease progressed: two abscesses formed, connected by sinus tracts. Steroids were considered but avoided — her surgeon, Dr. Kelly McLean of Christ Hospital in Cincinnati, had seen too many patients temporarily improve on steroids only to relapse once they tapered.
"It's kind of like a false sense of the disease actually getting resolved," Tami explains. "The symptoms often return."
The Invisible Organism
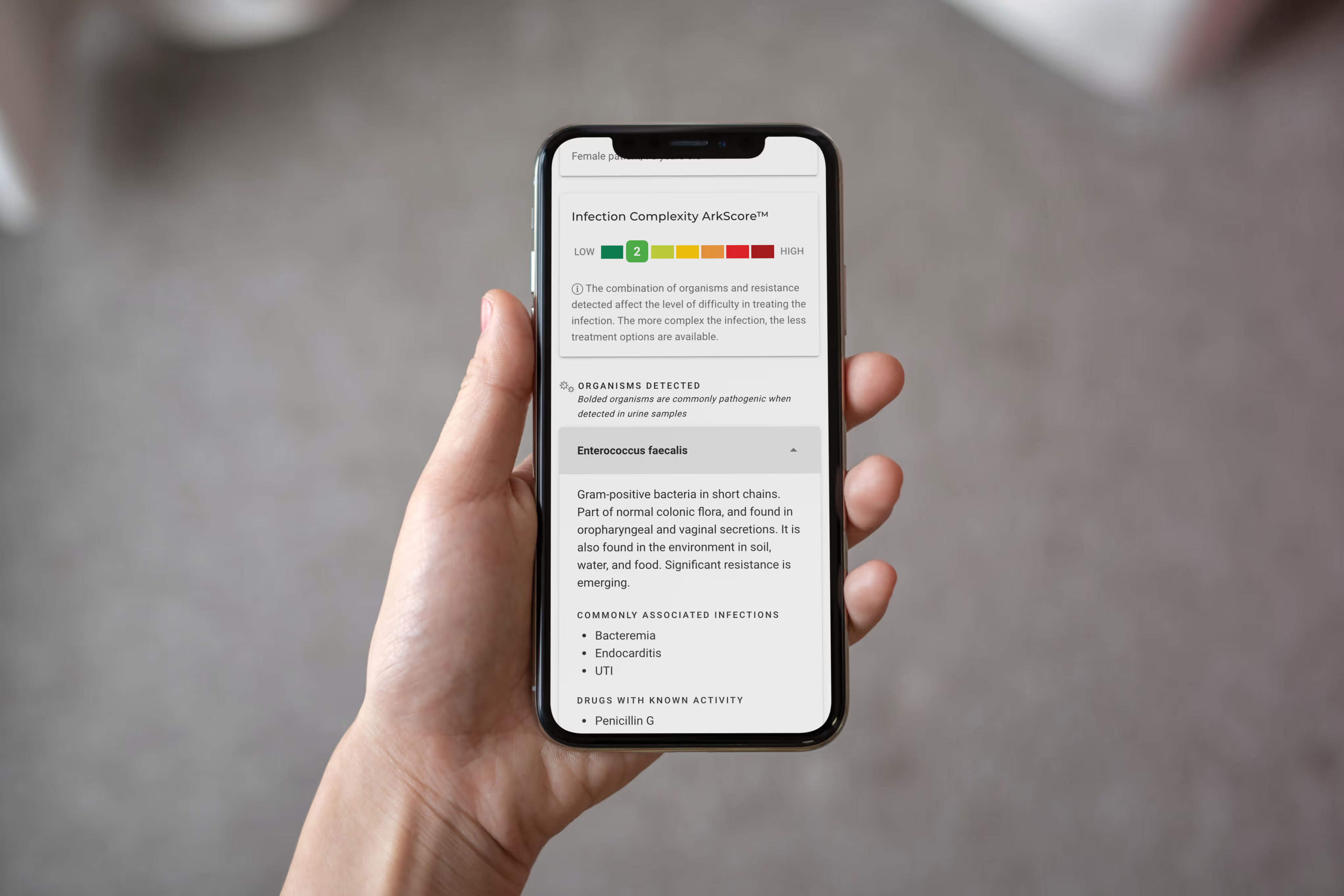
At the heart of Tami's story is a diagnostic truth that extends far beyond granulomatous mastitis: traditional laboratory methods have blind spots. Gram stains and cultures — the workhorses of microbiology — can fail to detect organisms like Corynebacterium kroppenstedtii. It doesn't always grow in culture. It's not always visible on a Gram stain. And when these tests come back negative, clinicians often conclude there is no infection present.
"These Gram stains and cultures often fail to detect this opportunistic microorganism," Tami explains. "It doesn't always grow in a culture. It's not always seen on a Gram stain."
Dr. Frenkel frames the broader problem: "The failure to detect a microbe is often viewed as there are no microbes there. So many times, physicians will see that a Gram stain or a culture has not grown an organism, and therefore, they rule out infection. However, there are lots of other techniques to detect microbes. And if you don't ask for this specific one, you can lead to a false negative."
When Tami eventually found the right diagnostic test — through her own research — she had to advocate for it with her surgeon. The test that identified the causative organism was not something her clinicians had initially ordered. The post-surgery confirmation test had to be sent to the Mayo Clinic.
"It's not really the patient's responsibility at that point to hunt down labs and to see what labs are available," Tami says. "But that's what I had to do."
The Erin Brockovich Moment
Perhaps the most remarkable part of Tami's story is the epidemiological detective work. After identifying the organism, she and her medical team traced her steps backward, where she'd been, what she'd done, when she'd done it. They traced the infection to a business trip to Connecticut, where she was exposed to contaminated public water from a hotel. A colleague on the same trip contracted an infection through his ankle from the same source, though in his case, the organism was never isolated. Her county tested her local water supply against the bacteria; it came back negative, further narrowing the source.
"We even took an Erin Brockovich-like step and figured out where I contracted the bacteria, which was contaminated public water. The contaminated public water found a natural point of entry through my breast ducts."
Why Doctors Miss What They Don't Know to Look For
The conversation turns systemic. Why do these diagnostic failures keep happening? Tami and Dr. Frenkel identify a convergence of forces:
Unfamiliarity with advanced testing. A surgical breast oncologist cannot be expected to know every molecular infectious disease test available for rare microbes. That expertise sits with infectious disease specialists — and many GM patients never see one.
Outdated training. Clinicians rely on what they learned in medical school, which may have been 20 years ago. The science of Corynebacterium and granulomatous mastitis has evolved significantly, but continuing education hasn't kept pace.
Time constraints. Tami's own surgeon contributed a striking admission to her book: "Due to her high clinical load, she just didn't have the time available to do the research that I did." Clinicians are managing patient volumes that leave minutes — not hours — per case.
The 80% problem. Dr. Frenkel notes that 80% of U.S. counties lack even one infectious disease physician. When generalists are left to manage complex infections without specialist support, the guessing game is inevitable.
"Clinicians are confined to a small window, sometimes minutes, to come up with conclusions on how to treat patients," Dr. Frenkel says. "That can lead to a trial-and-error approach where clinicians cycle through therapy they expect to work."
AI: From Threat to Superpower
Both Tami and Dr. Frenkel see AI as the inflection point. Patients are already using AI tools to research symptoms, interpret lab results, and arrive at their clinician's office with data. Tami envisions a world where even complex cases like hers can be triaged by AI that surfaces differentials human clinicians would never consider.
Dr. Frenkel goes further: the clinicians who embrace AI will become "super doctors."
"They can have a different outlook on the disease by inputting all this data into a system and getting differential diagnoses they may have never thought of. Clinicians who see this as a tool to enhance what they provide to their patients are going to really succeed."
Today, we don’t have to imagine a clinical decision support infrastructure that could have shortened Tami’s journey from seven months to weeks. Technology already exists that can interpret molecular results, flag organisms missed by cultures, and provide actionable guidance to generalists without infectious disease expertise.
The Stakes Are Higher Than One Patient
Tami's story is not just about granulomatous mastitis. It's about what happens in every hospital, every clinic, every urgent care when a clinician faces diagnostic uncertainty and reaches for an empiric antibiotic instead of a precise answer. The consequences cascade: ineffective treatment, disease progression, unnecessary surgeries, antibiotic resistance, C. diff risk, and patients left feeling gaslit by a system that couldn't find what was wrong.
"A lot of women who have gone through granulomatous mastitis often report feeling gaslit by their doctors. The management of the disease process itself is relentless. It is every single day of your life."
The episode closes with a shared conviction: responsible prescribing starts with better diagnostics, better collaboration, and the courage to admit that a negative culture doesn't mean the answer is "nothing."
🎧 Listen to the full conversation on Prescribe Responsibly — available on Spotify, Apple Podcasts, and YouTube.
This content is for educational purposes only and does not constitute medical advice.
Share this article
Blogs
Latest Blogs
Stay informed with our featured blogs.

When Negative Cultures Lie: The Hidden Cost of Diagnostic Blind Spots
When a culture comes back negative, most clinicians take it at face value. But a negative result is only as reliable as the method used to obtain it.
When a culture comes back negative, most clinicians take it at face value. But a negative result is only as reliable as the method used to obtain it.

When Antibiotics Miss the Mark Prescribe Responsibly · Episode 2
A conversation with Tami Burdick, Patient Advocate and Author of Diagnosis Detective: Curing Granulomatous Mastitis.
A conversation with Tami Burdick, Patient Advocate and Author of Diagnosis Detective: Curing Granulomatous Mastitis.
How Arkstone saves healthcare payers billions by eliminating unnecessary prescriptions
Most patients never know how often the right antibiotic is a guess. A faster, more confident answer changes everything for the patient, the clinician, and the hospital.
Most patients never know how often the right antibiotic is a guess. A faster, more confident answer changes everything for the patient, the clinician, and the hospital.
Customer Testimonials
Arkstone's solutions have transformed our patient care.
Take the Next Step, Try OneChoice®
No more guesswork. No more lookup tools. No more inaccuracies and inconsistencies. No more stopping to ask for directions.